Nemonapride
 | |
| Clinical data | |
|---|---|
| Trade names | Emilace (JP) |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | none |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | 75272-39-8 |
| PubChem (CID) | 4452 |
| IUPHAR/BPS | 983 |
| ChemSpider | 4297 |
| UNII |
Q88T5P3444 |
| KEGG |
D01468 |
| ChEBI | CHEBI:64217 |
| ChEMBL | CHEMBL20734 |
| ECHA InfoCard | 100.215.765 |
| Chemical and physical data | |
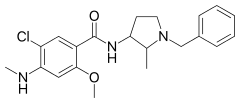
| Formula | C21H26ClN3O2 |
| Molar mass | 387.90 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Nemonapride (エミレース, Emilace (JP)) is an atypical antipsychotic approved in Japan for the treatment of schizophrenia. It was launched by Yamanouchi in 1991. Nemonapride acts as a D2 and D3 receptor antagonist, and is also a potent 5-HT1A receptor agonist. It has affinity for sigma receptors.
See also
References
This article is issued from Wikipedia - version of the 5/27/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.